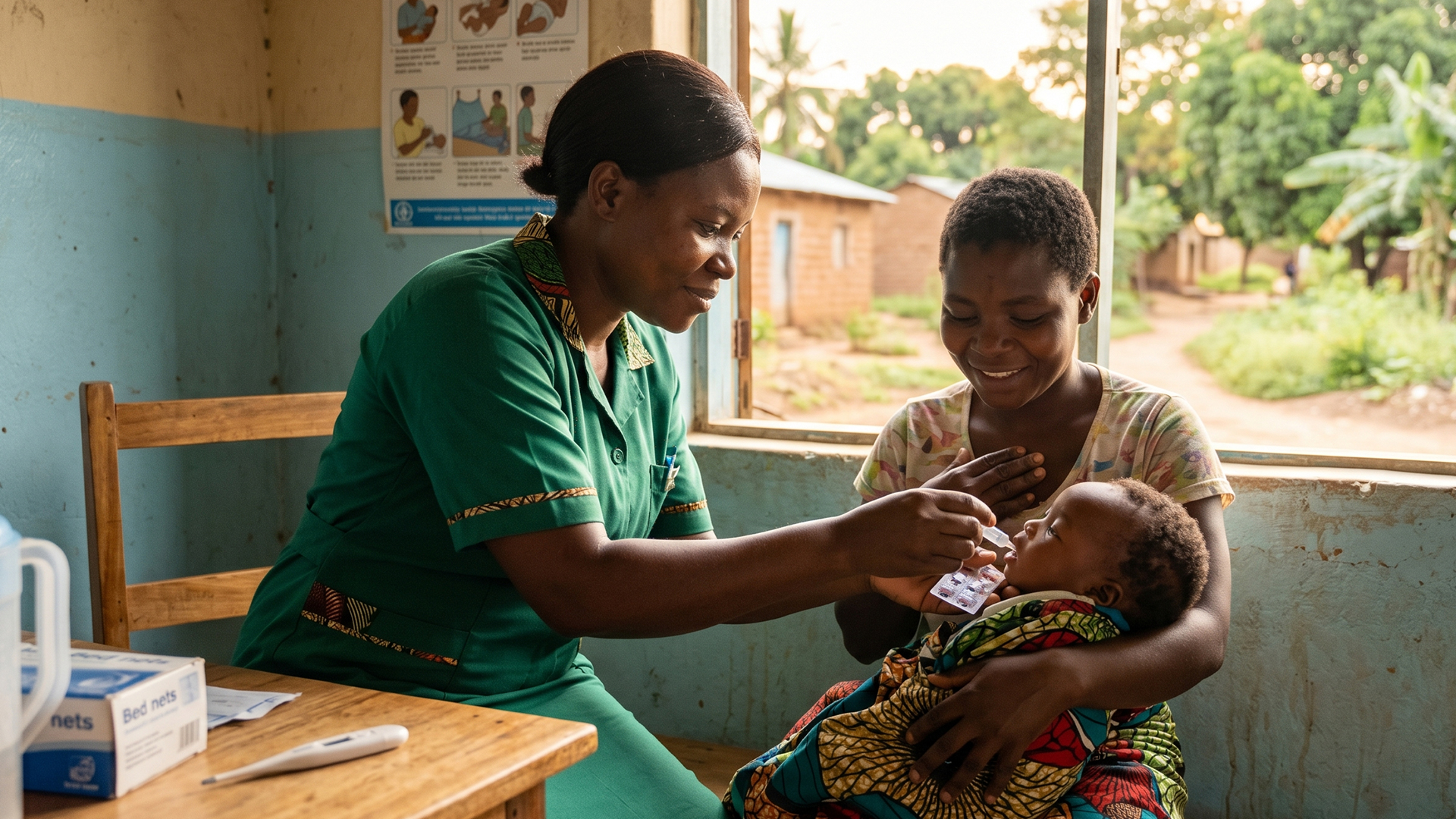
The World Health Organization (WHO) has approved the first malaria treatment specifically designed for newborns and very young infants, filling a long-standing gap in global healthcare.
Until now, babies infected with malaria were treated using medicines made for older children, which often required careful dose adjustments and carried a higher risk of complications. The new treatment has been specially formulated for infants weighing around 2 to 5 kilograms, making it safer and easier to use.
The drug, a low-dose version of artemether-lumefantrine, has been prequalified by WHO, meaning it meets international standards for quality, safety, and effectiveness. This approval allows it to be more widely supplied through global health programmes and national healthcare systems.
Health experts say the development is especially important for regions in Africa, where malaria continues to affect millions of people each year and young children remain the most vulnerable group. Newborns, in particular, have had very limited treatment options until now.
The infant-specific medicine is designed to reduce dosing errors and improve treatment outcomes, giving healthcare workers a more reliable option when treating very young patients.
Alongside the new drug, WHO also announced updated diagnostic tools for malaria. These tests are aimed at improving detection, especially in cases where certain parasite strains are harder to identify with older testing methods.
Also Read: Netanyahu says he was treated for cancer